Number of Lone Pairs and Bonding Pairs for CO3 2- (Carbonate ion) |

|
|
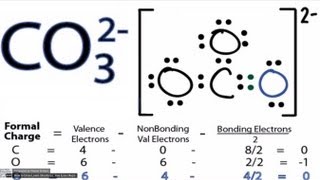
To determine the number of lone pairs and bonding pairs of electrons for CO3 2- we first need to draw as valid Lewis Structure. Once we have a Lewis Structure for CO3 2- then we can identify the lone and bonding pairs.
CO3 2- Lewis Structure:https://youtu.be/vheqX9W8bf4 CO3 2- Molecular Geometry: https://youtu.be/IIjnww-jwqQ Bonding pairs of electrons are the electrons between the atoms. These form the chemical bond and are shared between atoms. Often a pair of bonding electrons is represented by a line. Each line represents a pair of bonding electrons. Lone pairs of electrons are the remaining electrons around the atom. These are not between atoms and are not shared. The are important because they do occupy space and influence the shape of the molecule. There are no lone pairs of electrons on the Carbon (C) for CO3 2- (Carbonate ion). We often need to know the the number of lone pairs of electrons in a molecule like CO2 2- (as well as electrons involved in bonds) to determine the molecular geometry, calculate formal charges, and understand polarity and chemical reactivity. ---Learning Resources--- - How to Draw Lewis Structures: https://youtu.be/1ZlnzyHahvo - Lewis Structures Practice Video Worksheet: https://youtu.be/DQclmBeIKTc - Determining Formal Charge: https://youtu.be/vOFAPlq4y_k - Finding Valence Electrons (molecule): https://youtu.be/VBp7mKdcrDk - The Octet Rule: https://youtu.be/6Ecr7m-0E0E More help at http://www.breslyn.org |