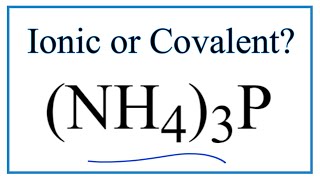Is (NH4)3P Ionic or Covalent/Molecular? |

|
|
To tell if (NH4)3P, Ammonium phosphide, is ionic or covalent (also called molecular) we we need to recognize NH4 + is a polyatomic ion and has a charge of 1+. The 1+ charge applies to the entire Ammonium ion.
The Nitride ion is P 3- overall ionic charge of 3-. When we have a the polyatomic ion NH4+ and a negative ion, like P 3-, the compound is considered ionic. Because the Ammonium ion, NH4 + and P 3- have opposite ionic charges, they attract and form an ionic bond. So in answer to our question (NH4)3P is an ionic compound. Do note that the bonds in the NH4 + polyatomic ion are covalent since they consist of only non-metals. But the overall compound, (NH4)3P is an ionic compound. --- Helpful Resources Metals, Non-Metals on the P- Table: https://youtu.be/OoooStZQHdA Ionic, Covalent, & Polar Covalent: https://youtu.be/OHFGXfWB_r4 Electronegativity for each element: https://en.wikipedia.org/wiki/Electronegativity Memorizing Polyatomic Ions: https://youtu.be/vepxhM_bZqk Finding Ionic Charge: https://youtu.be/N4N1Njh7nCo --- In general, ionic compounds: - form crystals. - have high melting points and boiling points. - are hard and brittle. - conduct electricity when dissolved in water. - as solids do not conduct electricity. For more chemistry help, see http://www.Breslyn.org. |