Pfizer: Full vaccine results indicate its Covid-19 vaccine is 95% effective |

|
|
Pfizer and BioNTech said Wednesday that a final data analysis found their coronavirus vaccine was 95% effective in preventing Covid-19 and, in addition, appeared to fend off severe disease. CNBC's Meg Tirrell reports. Subscribe to CNBC PRO for access to investor and analyst insights: https://cnb.cx/2Vtntx6
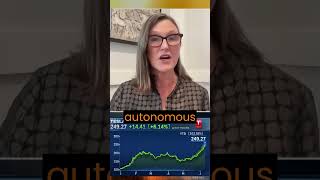
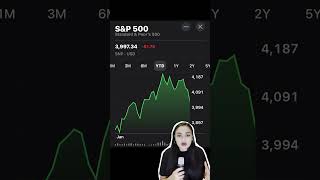
Pfizer and BioNTech said Wednesday that a final data analysis found their coronavirus vaccine was 95% effective in preventing Covid-19 and appeared to fend off severe disease. The vaccine, called BNT162b2, becomes highly effective against the virus 28 days after the first dose, and its effectiveness was consistent across all ages, races and ethnicities, the drugmakers said. Additionally, the elderly, who are seen as at high risk of severe illness from Covid-19, saw vaccine effectiveness of more than 94%, the companies said. “The final analysis underlines the results of the positive interim efficacy analysis announced on November 9,” BioNTech CEO Ugur Sahin said in a statement. “The data indicates that our vaccine ... is able to induce a high rate of protection against COVID-19 only 29 days after the first dose. In addition, the vaccine was observed to be well-tolerated in all age groups with mostly mild to moderate side effects, which may be due in part to the relatively low dose.” The vaccine also appeared to prevent severe disease in volunteers. Nine of the 10 severe cases of Covid were in the placebo group in the phase three trial, the companies said. They also saw no “serious” safety concerns, with most adverse events resolving shortly after vaccination. Pfizer’s shares jumped 3% in premarket trading, while BioNTech’s surged 6.2%. The final analysis evaluated 170 confirmed Covid infections among the late-stage trial’s more than 43,000 participants. The companies said 162 cases of Covid were observed in the placebo group versus eight cases observed in the group that received its two-dose vaccine. That resulted in an estimated vaccine efficacy of 95%, they said. The news comes more than a week after the companies announced that their vaccine was more than 90% effective and two days after Moderna said preliminary phase three trial data showed its vaccine was 94.5%. Both vaccines use messenger RNA, or mRNA, technology. It’s a new approach to vaccines that uses genetic material to provoke an immune response. A safe and effective vaccine is seen by investors and policymakers as a solution to get the global economy back on track after the pandemic wreaked havoc on nearly every country. The virus continues to spread rapidly, with more than 55.6 million cases worldwide and at least 1.33 million deaths as of Wednesday, according to data compiled by Johns Hopkins University. » Subscribe to CNBC TV: https://cnb.cx/SubscribeCNBCtelevision » Subscribe to CNBC: https://cnb.cx/SubscribeCNBC » Subscribe to CNBC Classic: https://cnb.cx/SubscribeCNBCclassic Turn to CNBC TV for the latest stock market news and analysis. From market futures to live price updates CNBC is the leader in business news worldwide. The News with Shepard Smith is CNBC’s daily news podcast providing deep, non-partisan coverage and perspective on the day’s most important stories. Available to listen by 8:30pm ET / 5:30pm PT daily beginning September 30: https://www.cnbc.com/2020/09/29/the-news-with-shepard-smith-podcast.html?__source=youtube%7Cshepsmith%7Cpodcast Connect with CNBC News Online Get the latest news: http://www.cnbc.com/ Follow CNBC on LinkedIn: https://cnb.cx/LinkedInCNBC Follow CNBC News on Facebook: https://cnb.cx/LikeCNBC Follow CNBC News on Twitter: https://cnb.cx/FollowCNBC Follow CNBC News on Instagram: https://cnb.cx/InstagramCNBC https://www.cnbc.com/select/best-credit-cards/ #CNBC #CNBCTV |