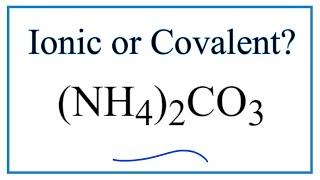How to Draw the Lewis Dot Structure for (NH4)2CO3: Ammonium carbonate |

|
|
A step-by-step explanation of how to draw the (NH4)2CO3 Lewis Dot Structure.
For (NH4)2CO3 we have an ionic compound and we need to take that into account when we draw the Lewis Structure. We’ll first draw the metal and put it in brackets with its charge on the outside (video: finding ionic charge: https://youtu.be/M22YQ1hHhEY). Next, we'll draw the Lewis Structure for the CO3 2- ion and add brackets. We put the two ions together to complete the Lewis structure for (NH4)2CO3. Note that (NH4)2CO3 is also called Ammonium carbonate. How to Draw the NH4+ Lewis Structure: https://youtu.be/LVZ5HzRLuzo How to Draw the CO3 2- Lewis Structure: https://youtu.be/vheqX9W8bf4 For a complete tutorial on drawing Lewis Structures, see my video: https://youtu.be/1ZlnzyHahvo For more practice, see https://youtu.be/DQclmBeIKTc To learn to find the valence electrons: https://youtu.be/VBp7mKdcrDk Note that is it more common to draw Lewis Structures for covalent (molecular) compounds where valance electrons are shared. In the case of ionic compounds, where we have a metal bonded to a non-metal (or group of non-metals), the Lewis diagram represents a formula unit. Many of these formula units make up a crystal lattice. So when we talk about the structure for (NH4)2CO3 we think of it together with other (NH4)2CO3 formula units in a crystal (NaCl is a good example of this: https://en.wikipedia.org/wiki/Sodium_chloride). Get more chemistry help at http://www.thegeoexchange.org/chemistry/bonding Drawing/writing done in InkScape. Screen capture done with Camtasia Studio 4.0. Done on a Dell Dimension laptop computer with a Wacom digital tablet (Bamboo). |