CO32- Lewis Structure (Carbonate Ion) |

|
|
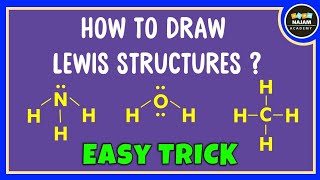
Hello Guys!
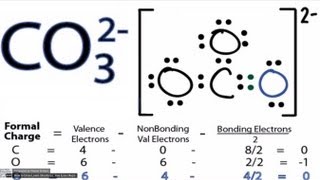
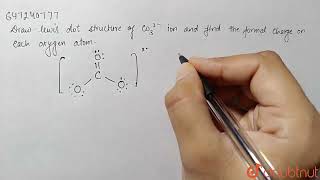
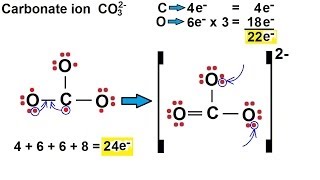
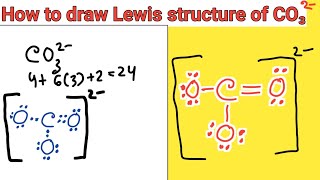
We are back with yet another video on Lewis structure and this time we are going to learn the Lewis structure of Carbonate Ion. It has a chemical formula of CO32-. The negative charge on this ion is because it accepts two extra valence electrons. In this video, we are going to look at the total number of valence electrons, bonds formed between the atoms and the formal charges on individual atoms. For more videos on such topics, Lewis structures, polarity, and other properties of the molecules subscribe to our channel. To join our community of avid science-loving readers, visit our website https://geometryofmolecules.com for more science-related videos, hit that subscribe button. Download all the slides in PDF format from here: https://jamboard.google.com/d/152VMNMjyKGq2Ra0D-0WEFVAFgdh0PAZCN_PvtHYO03A/viewer ► Below are the Tools we use to make our Videos more engaging : Best Video Editor Tool: https://tinyurlz.co/sfPr0 Best YouTube Marketing Tool: https://tinyurlz.co/yvyzQ #co32lewisstructure #CarbonateIon #CarbonateIonlewisstructure |