Writing the Formula for Carbon Dioxide |

|
|
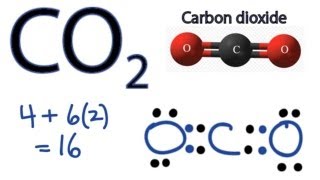
In this video we'll write the correct formula for Carbon Dioxide.
To write the formula for Carbon Dioxide we’ll use the Periodic Table and follow some simple rules. When we have a non-metal and a non-metal we have a molecular compound (sometimes called covalent). Molecular compounds are some of the simplest to name. Keys for Writing the Formulas for Molecular Compounds: - Write the element symbol for both elements. - Place a subscript after each element according to its prefix. Note: Don’t write the subscript '1'. --------- Prefixes mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10 --------- Caution: We only write "mono" for the second element in a molecular compound. So CO would be carbon monoxide. Monocarbon monooxide is incorrect. For a complete tutorial on naming and formula writing visit: http://www.thegeoexchange.org/chemistry/naming/resources/learning_naming.php or watch https://www.youtube.com/watch?v=zmdxMlb88Fs https://youtu.be/i_bcCmL3U28 Drawing/writing done in InkScape. Screen capture done with Camtasia Studio 4.0. Done on a Dell Dimension laptop computer with a Wacom digital tablet (Bamboo). |