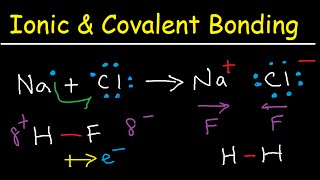
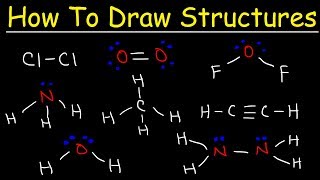
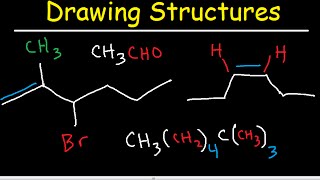
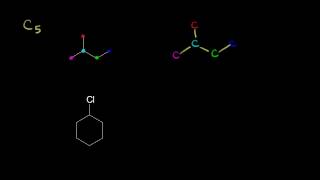
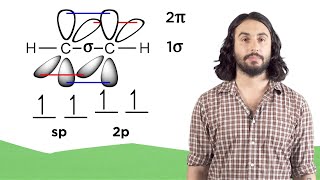
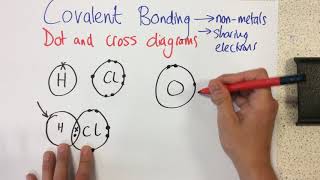
1. a. Draw the Lewis electron-dot structures for CO32-, CO2, and CO, including resonance structures… |

|
|
1. a. Draw the Lewis electron-dot structures for CO32-, CO2, and CO, including resonance structures where appropriate. 2) Which of the three species has the shortest C-O bond length? Explain your answer. 3) There are three compounds with the formula C2H2Cl2. Draw Lewis structures for these three compounds. Which of the three is non-polar?
Watch the full video at: https://www.numerade.com/ask/question/1-a-draw-the-lewis-electron-dot-structures-for-co32-co2-and-co-including-resonance-structures-where-appropriate-2-which-of-the-three-species-has-the-shortest-c-o-bond-length-explain-your-ans-86667/ Never get lost on homework again. Numerade is a STEM learning website and app with the world’s largest STEM video library. Join today and access millions of expert-created videos, each one skillfully crafted to teach you how to solve tough problems step-by-step. Join Numerade today at: https://www.numerade.com/signup/ #StructureandBonding |