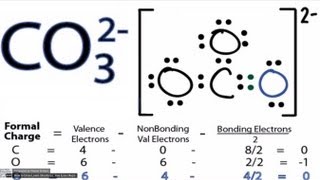
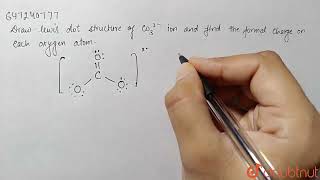
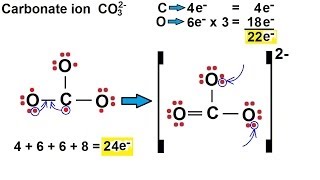
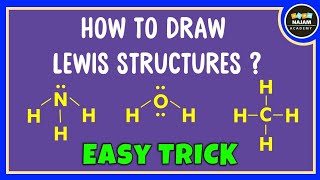
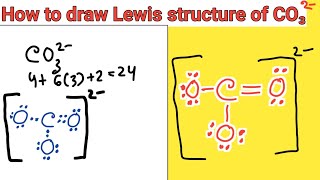
How to draw the Lewis structure of CO3 2- (Carbonate ion) |

|
|
Lewis structure of CO3 2- ion ||Lewis dot structure of co3 2- ||lewis dot structure of co3^2-
How to draw the Lewis Structure for CO3 2- (Carbonate ion)||Lewis structure of CO3 2- negative ion step by step Carbon follows to the octet rule due to the no presence of d orbital in the valence shell of of it - Carbon can handle 8 electrons. The Lewis Structure (Lewis Dot Diagram) for CO3 2-. 1. Count valence electrons for CO3 2- 2. Put least electronegative atom(C) in centre. 3. Put one electron pair in each bond 4. Fill outer atoms(O) with electrons first. 5. Move electrons so all atoms (esp. the centre one) has a full octet. |